February 24, 2026
Pedersen lab awarded NIH/NINDS R01 grant to study how to harness innate immunity to control HIV
The Pedersen lab has been awarded a new MPI NIH/NINDS R01 grant (5R01NS148274), titled: “Harnessing Innate Immunity to Control the Neuroinflammatory HIV Reservoir”, led by the Scintillon Research Institute, San Diego (co-PI: Irene Munk Pedersen, PhD.), and our colleagues at the University of California, Riverside (co-PI: Marcus Kaul, Ph.D.).
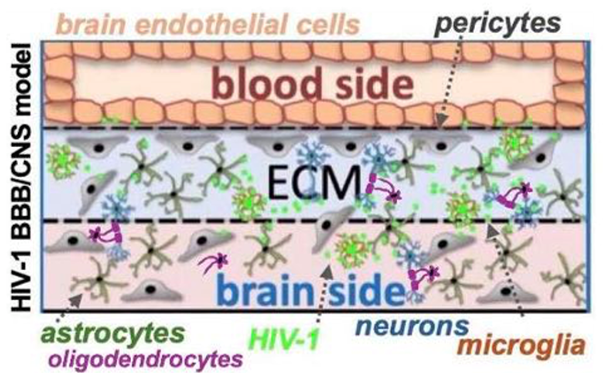
Perfused HIV-BBB/CNS model. 3D BBB-CNS culture systems with flow are generated employing the Mimetas platform and iPSC derived CNS cells and primary immune cells. One 3-lane chip carton representation. The top channel is seeded with brain endothelial cells against ECM in the middle channel, and CNS cells are seeded in the bottom channel. Leak-tight tubules form, CNS cells migrate into the ECM supporting BBB-formation and engage in CNS cellular networks. Immune cells and sera are introduced into the circulation (top channel) allowing for BBB transmigration and CNS infiltration, CSF is introduced into the bottom channel.
Human Immunodeficiency virus (HIV)-1 infects immune cells and affects all organs, including the brain, establishing viral reservoirs. If not appropriately treated, HIV infection leads to acquired immunodeficiency syndrome (AIDS) and associated neuro-cognitive impairment (NCI). However, even with combined retroviral therapy (cART), development of NCI occurs in about 50% of people with HIV and lifelong HIV-1 reservoirs persist. This project aims to harness the protective potential of interferon-β (IFNβ), against the formation of viral reservoirs and viral rebound and associated neurotoxicity using human tissue models of the brain.Our study aims at a better understanding of pathological mechanisms underlying HIV persistence and HIV-associated NCI to enable future therapeutic strategies. IFNβ is FDA-approved for the treatment of multiple sclerosis (MS), an inflammatory neurodegenerative autoimmune disease and this project will assess how IFNβ may be employed as a synergistic treatment with cART, which may offer a dual approach: cART for reducing viral replication and IFNβ for mitigating neuronal damage.